Chemical Kinetics and Nuclear ChemistryHard
Question
In a certain reaction, 10% of the reactant decomposes in one hour, 20% in two hours, 30% in three hours, and so on. The dimension of the velocity constant (rate constant) are
Options
A.Hr-1
B.Mol L-1 hr-1
C.L mol-1 s-1
D.Mol s-1
Solution
From the question, it is clear t1/2 ∝ a. Hence, zero order reaction. So, dimension of k = mol L-1 hr-1.
For zero order: t =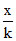 or k =
or k = 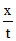
If t = t10% = 10 min, x = 10.
Then, k =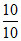 = 1 mol L-1 r-1
= 1 mol L-1 r-1
If t = t20% = 20 min, x = 20.
Then, k =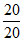 = 1 mol L-1 r-1
= 1 mol L-1 r-1
Similarly, for 30% and so on.
Thus, reaction is of zero order.
For zero order: t =
If t = t10% = 10 min, x = 10.
Then, k =
If t = t20% = 20 min, x = 20.
Then, k =
Similarly, for 30% and so on.
Thus, reaction is of zero order.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Which of the following represents the expression for 3/4th the life of a first-order reaction?...Consider a reaction aG + bH → Products. When concentration of both the reactants G and H is doubled, the rate incr...In gaseous reactions important for the understanding of the upper atmosphere, H2O and O react bimolecularly to form two ...emits 8α - particles and 6β - particles. The netron /proton ratio in the product nucleus is :...Reaction A + B → C + D + 38 Kcal has activation energy 20 Kcal. Activation energy for the reaction C + D → A...