Chemical BondingHard
Question
In piperidine 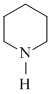 , the hybrid state assumed by N is
, the hybrid state assumed by N is
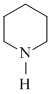 , the hybrid state assumed by N is
, the hybrid state assumed by N isOptions
A.sp
B.sp2
C.sp3
D.dsp2
Solution
Hybridisation of N = 1/2[5 + 3 + 0 - 0] = 4
hence sp3
hence sp3
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following is electron-deficient ?...During change of O2 to O2- ion, the electron adds on which one of the following orbitals?...The correct increasing order of $C - H(A)$, $C - O(B),C = O(C)$ and $C \equiv N(D)$ bonds in terms of covalent bond leng...The ionic radii (in (Ao)) of N3-, −O2- and F− are respectively :-...Given below are two statements:Statement I: Among $\left\lbrack Cu\left( {NH}_{3} \right)^{2 +},\left\lbrack Ni(en)_{3} ...