Chemical BondingHard
Question
The ionic radii (in (Ao)) of N3-, −O2- and F− are respectively :-
Options
A.1.71, 1.40 and 1.36
B.1.71, 1.36 and 1.40
C.1.36, 1.40 and 1.71
D.1.36, 1.71 and 1.40
Solution
Z e 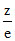
7N3− 7 10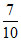 = 0.7
= 0.7
8O2− 8 10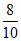 = 0.8
= 0.8
9F− 9 10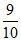 = 0.9
= 0.9
In case of isoelectronic species as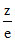 ratio increases ionic size decreases hence size order is
ratio increases ionic size decreases hence size order is
N3− > O2− > FΘ(1.71(Ao)) (1.40(Ao)) (1.36(Ao))
7N3− 7 10
8O2− 8 10
9F− 9 10
In case of isoelectronic species as
N3− > O2− > FΘ(1.71(Ao)) (1.40(Ao)) (1.36(Ao))
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
The number and type of bonds between two carbon atoms in CaC2 are :...Give the correct reaction :- (a) NaNO2 NO(g)(b) HNO3 N2O5(c) P4 + NaOH → PH3 + Na3PO4(d) (NH4)2Cr2O7 N2O + Cr2O3 +...Element M forms which types of sulphate :-...Assuming that Hund′s rule is violated, the bond order and magnetic nature of the diatomic molecule B2 is...Which one of the following compounds is a peroxide ?...