Chemical EquilibriumHard
Question
If K1 and K2 are respective equilibrium constants for the two reactions
XeF6(g) + H2O(g) ⇋ XeOF4(g) + 2HF(g)
XeO4(g) + XeF6(g) ⇋ XeOF4(g) + XeO3F2(g)
the equilibrium constant for the reaction
XeO4(g) + 2HF(g) ⇋ XeO3F2(g) + H2O(g)
XeF6(g) + H2O(g) ⇋ XeOF4(g) + 2HF(g)
XeO4(g) + XeF6(g) ⇋ XeOF4(g) + XeO3F2(g)
the equilibrium constant for the reaction
XeO4(g) + 2HF(g) ⇋ XeO3F2(g) + H2O(g)
Options
A.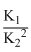
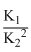
B.K1K2
C.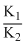
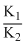
D.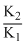
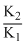
Solution
Reation (II) and reverse of reaction (i) gives the desired reaction hence
K = K2 ×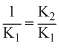
K = K2 ×
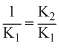
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The two equilibria, AB(aq) ⇋ A+(aq) + B-(aq) and AB(aq) + B-(aq) ⇋ AB2-(aq) are simultaneously maintained in...One mole of He (g, colourless) is added to the equilibrium mixture containing N2O4 (g, colourless) and NO2 (g, red-brown...The equilibrium constant for the reaction CO(g) + H2O(g) $\rightleftharpoons$CO2(g) + H2(g) is 3.0 at 500 K. In a 2.0 L ...Some quantity of water is contained in a container as shown in figure. As neon is added to this system at constant press...The following equilibria are given : N2 + 3H2 ⇋ 2NH3 K1 N2 + O2 ⇋ 2NO K2 H2 + O2 ⇋ H2O K3 The equilibr...