Chemical EquilibriumHard
Question
The two equilibria, AB(aq) ⇋ A+(aq) + B-(aq) and AB(aq) + B-(aq) ⇋ AB2-(aq) are simultaneously maintained in a solution with equilibrium constants, K1 and K2 respectively. The ratio of concentration of A+ to AB2- in the solution is :
Options
A.directly proportional to the concentration of B- (aq.).
B.inversely proportional to the concentration of B- (aq.).
C.directly proportional to the square of the concentration of B- (aq.).
D.inversely proportional to the square of the concentration of B- (aq.).
Solution
AB ⇋ A+ + B- AB + B- ⇋ AB2-
K1 =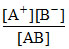 K2 =
K2 = 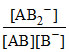
K1/K2 =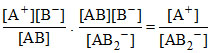 . [B-]2
. [B-]2
⇒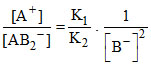
Therefore, (D) option is correct.
K1 =
K1/K2 =
⇒
Therefore, (D) option is correct.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The exothermic formation of ClF3 is represented by the equation: Cl2(g) + 3F2(g) ⇋ 2ClF3(g) ; ᐃrH = - 329 kJ...The correct relationship between free energy change in a reaction and the corresponding equilibrium constant K is...For the reaction X ⇋ 2Y and Z ⇋ P + Q occuring at two different pressure P1 and P2, respectively. The ratio ...The equilibrium $\text{SO}_2\text{Cl}_2(g) \rightleftharpoons \text{SO}_2(g) + \text{Cl}_2(g)$ is attained at $25^\circ\...Consider the following gaseous equilibrium in a closed container of volume "V" at T(K).$$P_{2}(\text{ }g) + Q_{2}(\text{...