Ionic EquilibriumHard
Question
The value Kp for the reaction :
2H2S(s) = 2H2(g) + S2(g) is 1.2 × 10-2 at 1065oC. The value for KC is :
2H2S(s) = 2H2(g) + S2(g) is 1.2 × 10-2 at 1065oC. The value for KC is :
Options
A.< 1.2 × 10-2
B.> 1.2 × 10-2
C.1.2 × 10-2
D.0.12 × 10-2
Solution
For the given reaction, 2H2S(g) = 2H2(g) + S2(g)
ᐃn = 3 - 2 = 1
Kc =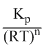
=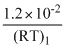
∴ KC < KP or KC < 1.2 × 10-2
ᐃn = 3 - 2 = 1
Kc =
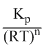
=
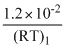
∴ KC < KP or KC < 1.2 × 10-2
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A + 2B ⇋ 2C + D, concentration of B was 1.5 time that of A, but the equilibriumconcentration of A and B are found ...The strength of an aqueous solution of I2 can be determined by titrating the solution with standard solution of:...Morphine (C17H19NO3) is administered medically to relieve pain. It is a naturally occurring base or alkaloid. What is th...A particular water sample has 136 ppm CaSO4. What percentage of water by mass must be evaporated in a container before s...Buffer solutions have constant acidity and alkalinity because...