ElectrochemistryHard
Question
Given the ionic conductance of 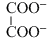 , K+, and Na+ are 74, 50 and 73 cm2 ohm-1 eq-1, respectively. The equivalent conductance at infinite dilution of the salt
, K+, and Na+ are 74, 50 and 73 cm2 ohm-1 eq-1, respectively. The equivalent conductance at infinite dilution of the salt 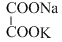 is
is
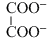 , K+, and Na+ are 74, 50 and 73 cm2 ohm-1 eq-1, respectively. The equivalent conductance at infinite dilution of the salt
, K+, and Na+ are 74, 50 and 73 cm2 ohm-1 eq-1, respectively. The equivalent conductance at infinite dilution of the salt 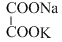 is
isOptions
A.197 cm2 ohm-1 eq-1
B.172 cm2 ohm-1 eq-1
C.135.5 cm2 ohm-1 eq-1
D.160.5 cm2 ohm-1 eq-1
Solution
Total charge = 2
Number of equivalent of ion =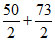
∴ Eq of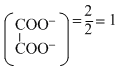
Eq of Na+ =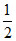 , Eq of K+ =
, Eq of K+ = 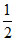
∴ λoeq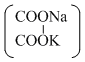
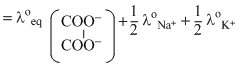
= 74 +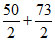 = 135.5
= 135.5
Number of equivalent of ion =
∴ Eq of
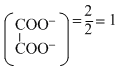
Eq of Na+ =
∴ λoeq
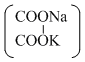
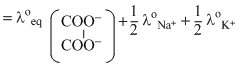
= 74 +
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The copper anode of a cell containing silver nitrate solution weighs 60.0 g. After passing current for some time, it is ...Which cannot displace hydrogen from its compound ?...M(s) → Mn+ (aq) + ne-Choose the correct statement(s)....For the cell: Ni|Ni2+||Cu2+|Cu; E° = 0.77 V. By which of the following activity, Ecell will increase?...The standard reduction potentials for Zn2+ / Zn,  Ni2+ / Ni and Fe2+ / Fe are - 0.76, - 0.23 and - 0.44 V respectiv...