ElectrochemistryHard
Question
Time required to deposit one millimole of aluminum metal by the passage of 9.65 amperes through aqueous solution of aluminum ion is
Options
A.30 s
B.10 s
C.30,000 s
D.10,000 s
Solution
l3+ + 3e- → Al
1mole requires = 3 × 96500 coulomb
1 millimole (10-3mol)requires
= 3 × 96500 × 10-3C
we know that, Q = i t
or t =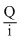
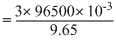
=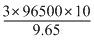 30 sec.
30 sec.
1mole requires = 3 × 96500 coulomb
1 millimole (10-3mol)requires
= 3 × 96500 × 10-3C
we know that, Q = i t
or t =
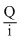
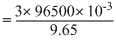
=
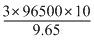 30 sec.
30 sec.Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The temperature coefficient, of the emf i.e. = - 0.00065 volt. deg-1 for the cell Cd | CdCl2 (1M) | AgCl (s) | Ag at 25o...Which cannot displace hydrogen from its compound ?...When galvanic cell started, with passage of time...At the Nangal fertilizer plant in Punjab, hydrogen is produced by the electrolysis of water. The hydrogen is used for th...Lactic acid, HC3H5O3, produced in 1 g sample of muscle tissue was titrated using phenolphthalein as indicator against OH...