ElectrochemistryHard
Question
The temperature coefficient, of the emf i.e. 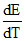 = - 0.00065 volt. deg-1 for the cell Cd | CdCl2 (1M) | AgCl (s) | Ag at 25oC. Calculate the entropy changes ᐃS298K for the cell reaction, Cd + 2AgCl → Cd++ + 2Cl- + 2Ag
= - 0.00065 volt. deg-1 for the cell Cd | CdCl2 (1M) | AgCl (s) | Ag at 25oC. Calculate the entropy changes ᐃS298K for the cell reaction, Cd + 2AgCl → Cd++ + 2Cl- + 2Ag
Options
A.- 105.5 JK-1
B.- 150.2 JK-1
C.- 75.7 JK-1
D.- 125.5 JK-1
Solution
ᐃS298 = n.F.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
If same quantity of electricity is passed through three electrolytic cells containing FeSO4, Fe2(SO4)3 and Fe(NO3)3, the...Which statement is correct....The standard reduction potentials of Cu2+|Cu and Cu2+|Cu+ are 0.337 V and 0.153 V, respectively. The standard electrode ...At the Nangal fertilizer plant in Punjab, hydrogen is produced by the electrolysis of water. The hydrogen is used for th...A metal having negative reduction potential, when dipped in the solution of its own ions, has a tendency to...