Gaseous StateHard
Question
An element (atomic mass 100g/mol) having BCC structure has unit cell edge 400 pm. the density of element is (No. of atoms in BCC, Z = 2).
Options
A.2.144g/cm3
B.7.289 g/cm3
C.5.188 g/cm3
D.10.376 g/cm3
Solution
Mass of two atoms = 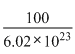 × 2g
× 2g
=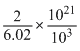 kg
kg
Volume of cell = (4 × 10-10)3 = 64 × 10-30
Density =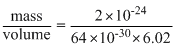
Density =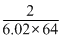 × 106 kg/m3
× 106 kg/m3
=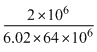 gm/cc
gm/cc
=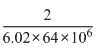 gm/cc = 5.188gm/cc
gm/cc = 5.188gm/cc
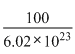 × 2g
× 2g=
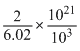 kg
kgVolume of cell = (4 × 10-10)3 = 64 × 10-30
Density =
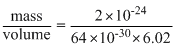
Density =
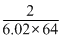 × 106 kg/m3
× 106 kg/m3=
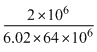 gm/cc
gm/cc=
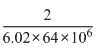 gm/cc = 5.188gm/cc
gm/cc = 5.188gm/ccCreate a free account to view solution
View Solution FreeMore Gaseous State Questions
As the temperature is raised from 20oC to 40oC, the average kinetic energy of neon atoms changes by a factor of which of...By what factor does the average velocity of a gaseous molecule increase when the temperature (in Kelvin) is doubled ?...A reversible cyclic process for an ideal gas is shown below. Here, P , V and T are pressure , Volume and temperature , r...A certain volume of argon gas (Mol. Wt. = 40) requires 45 s to effuse through a hole at a certain pressure and temperatu...50 mL of each gas A and of gas B takes 200 and 150 s respectively for effusing through a pin hole under the similar cond...