Gaseous StateHard
Question
Consider the following reactions in which all the reactants are products are in gaseous state :-
2PQ ⇋ P2 + Q2 ; K1 = 2.5 × 105
PQ +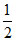 R2 ⇋ PQR; K2 = 5 × 10-3
R2 ⇋ PQR; K2 = 5 × 10-3
The value of K3 for the equilibrium
2PQ ⇋ P2 + Q2 ; K1 = 2.5 × 105
PQ +
The value of K3 for the equilibrium
Options
A.2.5 × 10-3
B.2.5 × 103
C.1.0 × 10-5
D.5 × 103
More Gaseous State Questions
Three footballs are respectively filled with nitrogen, hydrogen and helium. If the leaking of the gas occurs with time f...Temperature at which r.m.s. speed of O2 is equal to that of neon at 300 K is :...Assuming that O2 molecule is spherical in shape with radius 2(Ao), the percentage of the volume of O2 molecules to the t...A hypothetical gaseous element having molecular formula Mx may be changed to another gaseous allotrope having molecular ...For real gases van der Waals equation is written as (V - nb) = nRT where ′a′ and ′b′ are van der...