Chemical BondingHard
Question
Which of the following has the highest dipole moment ?
Options
A.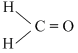
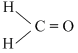
B.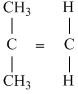
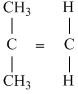
C.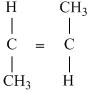

D.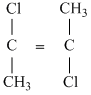

Solution
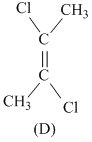
Greater the difference in electronrgativity between the two atoms, larger will be polarity and hence dipole moment. Thus (a) Has maximum dipole moment.
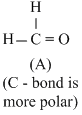
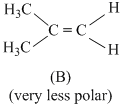
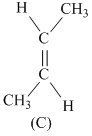
Symmertrical molecules (μ = 0)
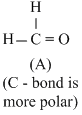



Symmertrical molecules (μ = 0)
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Using MO theory predict which of the following species has the shortest bond length ?...A gas X (mol. wt. = 100 g) decomposes into Y according to the following equation :X(g) ⇋ 2Y(g)At equillibrium, vap...Predict the correct order among the following:...The pair of bases in DNA are held together by:-...Element M forms which types of sulphate :-...