Chemical BondingHard
Question
Using MO theory predict which of the following species has the shortest bond length ?
Options
A.O22+
B.O2+
C.O2-
D.O22-
Solution
Bond length 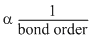
Bond order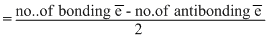
Bond orders of O2+, O2-, O2-2 and O2+2 are respectively
2.5, 1.5, 1 and 3.
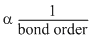
Bond order
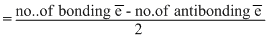
Bond orders of O2+, O2-, O2-2 and O2+2 are respectively
2.5, 1.5, 1 and 3.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
The major product formed in the following reaction is : →HeatHI...In which of the following ion, the hybrid state of halogen is sp3 :-...The species having bond order different from that in CO is...Which of the two ions from the list given below, have the geometry that is explained by the same hybridisation of orbita...Consider the plots, gives below, for the types of reaction:- nA → B + C (I) (II) (III)These plots respectively cor...