Chemical BondingHard
Question
A gas X (mol. wt. = 100 g) decomposes into Y according to the following equation :
X(g) ⇋ 2Y(g)
At equillibrium, vapour density and total pressure is found 37.5 and 2 atm respectively. The KP for the reaction is :
X(g) ⇋ 2Y(g)
At equillibrium, vapour density and total pressure is found 37.5 and 2 atm respectively. The KP for the reaction is :
Options
A.4
B.2
C.3
D.1
Solution
X(g) ⇋ 2Y(g)
a
a-aα 2aα
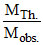 = 1 + α ⇒
= 1 + α ⇒ 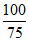 = 1 + α ⇒ α =
= 1 + α ⇒ α = 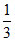
KP =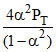 = 1
= 1
a
a-aα 2aα
KP =
Create a free account to view solution
View Solution Free