Atomic StructureHard
Question
Find the frequency of light that corresponds to photons of energy 5.0 × 10-5 erg
Options
A.7.5 × 10-21 sec-1
B.7.5 × 10-21 sec
C.7.5 × 1021 sec-1
D.7.5 × 1021 sec
Solution
Using E = hv, we get
v =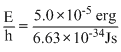
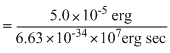
[∵ 1 J = 107 erg]
= 7.54 × 1021 sec-1
v =
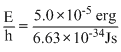
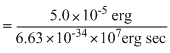
[∵ 1 J = 107 erg]
= 7.54 × 1021 sec-1
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
If the principal quantum number n = 6, the correct sequence of filling of electrons will be :...The I.P. of an atom is 16 eV. What is the value of first excitation potential :-...The uncertainty in position and velocity of the particle are 0.1 nm and 5.27 × 10-27 ms-1 respectively then the mas...The wavelength of a spectral lime for an electronic transition is inverssly related to :...A molecule XY2 contains two σ, two π - bonds and one lone pair of electron in the valence shell of X. The arr...