Atomic StructureHard
Question
The I.P. of an atom is 16 eV. What is the value of first excitation potential :-
Options
A.10.2 eV
B.12 eV
C.14 eV
D.16 eV
Solution
E∞ - E1 = 16 eV ⇒ E1 = -16 eV
E2 =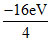 = - 4 eV
= - 4 eV
Ist excitation potential = E2 - E1
= - 4 eV - (-16 eV) = 12 eV
E2 =
Ist excitation potential = E2 - E1
= - 4 eV - (-16 eV) = 12 eV
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Maximum number of electrons in a subshell with l = 3 and n = 4 is...The percentage of p-character in the orbitals forming P-P bonds in P4 is...The number of possible spectral lines in the bracket series in hydrogen spectrum, when electrons present in the ninth ex...Which is/are correct for sodium atom in the ground state?...In an oil drop experiment, the following charges (in arbitrary units) were found on a series of oil droplets 4.5 × 10−18...