SolutionHard
Question
A solution is prepared by dissolvint 24.5 g of sodium hydroxide in distilled water to give 1L solution. The molarityof NaOH in the solution is
(Give that molar mass of NaOH = 40.0 g mol-1)
(Give that molar mass of NaOH = 40.0 g mol-1)
Options
A.0.2450 M
B.0.6125 M
C.0.9800 M
D.1.6326 M
Solution
Given WNaOH = 24.5g
No. of moles of NaOH =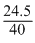 moles
moles
= 0.6125 moles
∴ Molarity of solution
=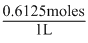 = 0.6125 M
= 0.6125 M
No. of moles of NaOH =
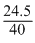 moles
moles = 0.6125 moles
∴ Molarity of solution
=
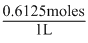 = 0.6125 M
= 0.6125 MCreate a free account to view solution
View Solution FreeMore Solution Questions
The process of getting fresh water from sea water is known as...At 298 K , the mole percentage of $N_{2}(\text{ }g)$ in air is $80\%$. Water is in equilibrium with air at a pressure of...A current of 2.6 ampere was passed through CuSO4 solution for 380 sec. The amount of Cu deposited is (at. wt. of Cu = 63...The vapour pressure of a solution of a non-volatile solute B in a solvent A is 95% of the vapour pressure of the solvent...The degree of dissociation (α) of a weak electrolyte, Ax By is related to van′t Hoff factor (i) by the expres...