SolutionHard
Question
0.01 M solution of KCl and BaCl2 are prepared in water. The freezing point of KCl is found to be 2oC.
Options
A.- 3oC
B.+ 3oC
C.- 2oC
D.- 4oC
Solution
ᐃTf ∝ i
∴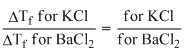
∴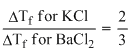 (∵ ᐃTf for KCl; = 2)
(∵ ᐃTf for KCl; = 2)
∴ ᐃTf for BaCl2 =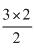 = 3
= 3
∴ Freezing point of BaCl2 = - 3oC
∴
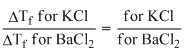
∴
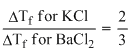 (∵ ᐃTf for KCl; = 2)
(∵ ᐃTf for KCl; = 2)∴ ᐃTf for BaCl2 =
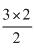 = 3
= 3∴ Freezing point of BaCl2 = - 3oC
Create a free account to view solution
View Solution FreeMore Solution Questions
The vapour pressure of water depends upon :...20 g of a binary electrolyte (mol. mass = 100) are dissolved in 500 g of water. The freezing point of the solution is -0...The vapour pressure of a solution of a non-volatile and non-electrolyte solute in a solvent is 95% of the vapour pressur...The molal depression constant for four liquids P, Q, R and S, respectively, are 1.84, 2.20, 3.15 and 3.92. If 0.1 m urea...An aqueous solution of a solute AB has b.p. of 101.08oC (AB is 100% ionised at boiling point of the solution) and freeze...