SolutionHard
Question
The vapour pressure of pure benzene at a certain temperature is 0.850 bar. A non-volatile non-electrolyte solid weighing 0.5 g is added to 39.0 g of benzene (molar mass 78g/mol). The vapour pressure of the solution then is 0.845 bar. What is the molecur mass of the solid substance ?
Options
A.58
B.180
C.170
D.145
Solution
pobenzene = 0.850bar ; Wbenzene = 39.0 g.
M. W. = 78g g/mol.
pobenzene = 0.850bar w = 0.5 g.
Let n be the. of, oles of non volatile solid; w and m be the weight of solid and molecular mass respectively.
Using,
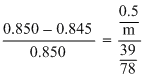
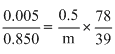
m = 170
= 170
M. W. = 78g g/mol.
pobenzene = 0.850bar w = 0.5 g.
Let n be the. of, oles of non volatile solid; w and m be the weight of solid and molecular mass respectively.
Using,

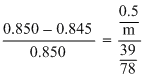
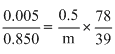
m
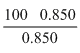 = 170
= 170Create a free account to view solution
View Solution FreeMore Solution Questions
After removing the hard shell of an egg by dissolving in dilute HCl, a semipermeable membrane can be visible. If such an...Consider the following reactions.(Hot solution)In the above reactions, and X are respectively....18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution at 100oC ...Gold number of haemoglobin is 0.03. Hence, 1000 mL of gold sol will require haemoglobinso that gold is not coagulated by...For a dilute solution having molality m of a given solute in a solvent of mol.wt. M, b.pt. Tb and heat of vaporisation p...