SolutionHard
Question
For a dilute solution having molality m of a given solute in a solvent of mol.wt. M, b.pt. Tb and heat of vaporisation per mole ᐃH ; 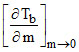 is equal to :
is equal to :
Options
A.Molal elevation constant of solvent
B.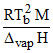 ; where M in kg ᐃvap H and R in J mol-1
; where M in kg ᐃvap H and R in J mol-1
C.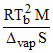 ; where M in kg ᐃvapS and R in J mol-1
; where M in kg ᐃvapS and R in J mol-1
D.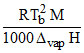 ; where M in g ; R and ᐃvapH expressed in same unit of heat.
; where M in g ; R and ᐃvapH expressed in same unit of heat.
More Solution Questions
Van′t Hoff factor is :...Which of the following is not a characteristic property of the polar liquids?...The molar mass of substance forming 7.0% (by mass) solution in water which freezes at −0.93°C, is (The cryoscopic consta...Vapour density of PCl5 (g) dissociating into PCl3(g) and Cl2(g) is 100. Hence, van′t Hoff factor for the case : PC...H2S on passing through ammonical solution gives white ppt. The white ppt. is of :-...