SolutionHard
Question
The normality of orthophosphoric acid having purity of 70%by weight and specific gravity 1.54 is :The normality of orthophosphoric acid having purity of 70%by weight and specific gravity 1.54 is :
Options
A.11 N
B.22 N
C.33 N
D.44 N
Solution
70% by weight means Wt. of solute = 70 g
Wt of solution = 100g
∴ V∝ of solution =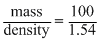
We know that
N =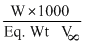
=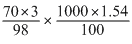 = 33 N
= 33 N
Wt of solution = 100g
∴ V∝ of solution =
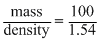
We know that
N =
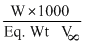
=
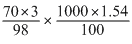 = 33 N
= 33 NCreate a free account to view solution
View Solution FreeMore Solution Questions
n aqueous solution of NaCl shows the An aqueous solution point of water equal to 0.372 K. The boiling point of BaCl2 sol...When the depression in freezing point is carried out, the equilibrium exist between...Vapour density of PCl5 (g) dissociating into PCl3(g) and Cl2(g) is 100. Hence, van′t Hoff factor for the case : PC...Barium ions, CN- and Co2+ form an ionic complex. If that complex is supposed to be 75% ionised in water with vant Hoff f...When 36.0 g of a solute having the empirical formula CH2O is dissolved in 1.20 kg of water, the solution freezes at −0.9...