SolutionHard
Question
n aqueous solution of NaCl shows the An aqueous solution point of water equal to 0.372 K. The boiling point of BaCl2 solution of same molarity will be
[Kf(H2O) = 1.86K kg mol-1; Kb(H2O) = 0.52K kg mol-1]
[Kf(H2O) = 1.86K kg mol-1; Kb(H2O) = 0.52K kg mol-1]
Options
A.100.52oC
B.100.104oC
C.101.56oC
D.100.156oC
Solution
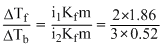
ᐃTf = 0.372. Hence, ᐃTb = 0.156oC
i.e., Tb = 100.156oC
Create a free account to view solution
View Solution FreeMore Solution Questions
18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution at 100oC ...Identify the false statement :...The freezing point among the following equimolal aqueous solutions, will be highest for :...In the depression of freezing point experiment, it is found that the :...If P0 and PS, are the V.P. of solvent and its solution respectively and N1 and N2 are the mole fraction of solvent and s...