SolutionHard
Question
Vapour density of PCl5 (g) dissociating into PCl3(g) and Cl2(g) is 100. Hence, van′t Hoff factor for the case :
PCl5(g) ⇋ PCl3(g) + Cl2(g) is
PCl5(g) ⇋ PCl3(g) + Cl2(g) is
Options
A.1.85
B.3.70
C.1.085
D.1.0425
Solution
PCl5 ⇋ PCl3 + Cl2
i = 1 + (y - 1) x = (1 + x)
x =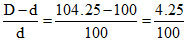 = 0.0425
= 0.0425
∴ i = 1 + x = 1.0425
(D = theoretical VD =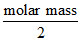
d = experimental VD)
i = 1 + (y - 1) x = (1 + x)
x =
∴ i = 1 + x = 1.0425
(D = theoretical VD =
d = experimental VD)
Create a free account to view solution
View Solution FreeMore Solution Questions
Consider following solutions :I : 1 M aqueous glucose solution II : 1M aqueous sodium chloride solution III : 1M aqueous...The osmotic pressures of 0.010 M solutions of KI and of sucrose (C12H22O11) are 0.432 atm and 0.24 atm respectively. The...An ionic compound that attracts atmospheric water so strongly that a hydrate is formed is said to be :...An aqueous solution of 10% NaCl (consider ideal behaviour of the solution) is cooled. It will allow some...All of the water in a 0.20 M solution of NaCl was evaporated and a 0.150 mol of NaCl was obtained. What was the original...