SolutionHard
Question
The molar concentration of 20 g of NaOH present in 5 liter of solution is
Options
A.0.1 moles/litre
B.0.2 moles/litre
C.1.0 moles/litre
D.2.0 moles/litre
Solution
Molar concentration = 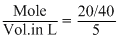
=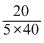 = 0.1 mole/litre.
= 0.1 mole/litre.
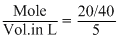
=
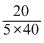 = 0.1 mole/litre.
= 0.1 mole/litre.Create a free account to view solution
View Solution FreeMore Solution Questions
80 mL of a hydrocarbon on mixing with 264 mL of oxygen in a closed U-tube undergoes complete combustion. The residual ga...A solution of glucose ( C6H12O6) is isotonic with 4 g of urea ( NH2-CO-NH2) per liter of solution. The concentration of ...An ideal solution is obtained by dissolving n moles of non-volatile, non-electrolyte solute in N moles of solvent. If th...The depression of freezing points of 0.05 molal aqueous solution of the following compounds are measured.1. NaCl 2. K2SO...Liquids P and Q form an ideal solution. The vapour pressures of pure liquids P and Q at 80°C are 300 mm and 100 mm of Hg...