SolutionHard
Question
A solution of glucose ( C6H12O6) is isotonic with 4 g of urea ( NH2-CO-NH2) per liter of solution. The concentration of glucose is :
Options
A.4 g/l
B.8 g/l
C.12 g/l
D.14 g/l
Solution
(a) Isotonic solution has same conc.
π1 = π2 C1 = C2 n1 = n2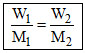
So,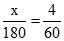 ⇒ x = 12 g Ans.
⇒ x = 12 g Ans.
π1 = π2 C1 = C2 n1 = n2
So,
Create a free account to view solution
View Solution FreeMore Solution Questions
Phenol dimerizes in benzene. If the observed molecular mass of phenol in solution is 120, then its degree of dimerizatio...At 323 K, the vapour pressure in millimeters of mercury of a methanol-ethanol solution is represented by the equation p ...Three solutions of HCl having normality 12 N, 6 N and 2 N are mixed to obtain a solution of 4 N normality. Which among t...At a particular temperature and at a pressure of 1 atm, the mass and volume of O3 that can be dissolved in 1 L of H2O is...The latent heat of vaporization of a liquid of molar mass, 80 g/mol and boiling point, 127°C is 8 kcal/mol. The ebullios...