Mole ConceptHard
Question
To neutralize completely 20 mL of 0.1 M aqueous solution of phosphorous acid (H3PO3), the volume of 0.1 M aqueous KOH solution required is
Options
A.10 mL
B.60 mL
C.40 mL
D.20 mL
Solution
No. of gm equivalents of phosphorous acid
= No. of gm equivalents of KOH
20 × 0.1 × 2 (n = factor) = 0.1 × V
= 0.1 × V
V =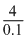 = 40 ml
= 40 ml
= No. of gm equivalents of KOH
20 × 0.1 × 2 (n = factor) = 0.1 × V
= 0.1 × V
V =
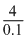 = 40 ml
= 40 mlCreate a free account to view solution
View Solution FreeMore Mole Concept Questions
In which of the following pairs, 10 g of each have an equal number of molecules ?...What mass of carbon disulphide CS2 can be completely oxidized to SO2 and CO2 by the oxygen liberated when 325 g of Na2O2...A sample of oleum is labelled as 112%. In 200 g of this sample, 18 g water is added. The resulting solution will contain...A 2 m long tube closed at one end is lowered vertically into water until the closed end is flushed with the water surfac...Which of the following is not a characteristic of chemisorption :-(a) Adsorption is irreversible(b) It results into mult...