Mole ConceptHard
Question
Mole fraction of the component A in vapour phase is x1 and mole fraction of component A in liquid mixture is x2 (PoA = vapour pressure of pure A; PoB = vapour pressure of pure B) then total vapour pressure of the liquid mixture is :-
Options
A.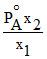
B.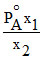
C.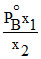
D.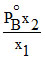
Solution
P′A = X2PoA , P′A = X1Pm
⇒ X1Pm = X2PAo ⇒ Pm =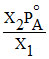
⇒ X1Pm = X2PAo ⇒ Pm =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
In Duma′s method of estimation of nitrogen 0.35 g of an organic compound gave 55 mL of nitrogen collected at 300 K...For the complete combustion of 0.2 moles of the sugar, how many moles of oxygen is required ?...One mole of the complex compound Co(NH3)5Cl3, gives 3 moles of ions on dissolution in water. One mole of the same comple...10 L of hard water required 0.56 g of lime (CaO) for removing hardness. Hence, temporary hardness in ppm (part per milli...Which of the following statements is/are correct ?20.0 mL of 6.0 M HCl is mixed with 50.0 mL of 2.0 M Ba(OH)2, and 30 mL...