Atomic StructureHard
Question
If e = 1.60206 × 10-19 C,
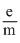 = 1.75875 × 1011C kg-1
= 1.75875 × 1011C kg-1
then mass of electron is
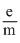 = 1.75875 × 1011C kg-1
= 1.75875 × 1011C kg-1then mass of electron is
Options
A.7.5678 × 10-31kg
B.9.1091 × 10-31kg
C.11.2531 × 10-31kg
D.13.0513 × 10-31kg
Solution
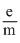 = 1.75875 × 1011
= 1.75875 × 1011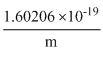 = 1.75875 × 1011
= 1.75875 × 1011m =
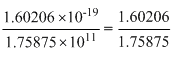 × 10-30
× 10-30= 9.1091 × 10-31kg
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
If the nitrogen atom had electronic configuration 1s7, it would have energy lower than that of the normal ground state c...Sodium atoms emit a spectral line with a wavelength in the yellow, 589.6 nm. What is the approximate difference in ener...In H-atom, the wave number ratio is 108: 7 is for...Wavelength of radiations emitted when an electron jumps from a state A to C is 3000 and it is 6000 when the electron jum...The ratio of specific charge (e/m) of a proton and that of an a-particle is...