Atomic StructureHard
Question
Wavelength of radiations emitted when an electron jumps from a state A to C is 3000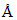 and it is 6000
and it is 6000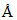 when the electron jumps from state B to C. Wavelength of the radiations emitted when an electron jumps from state A to B will be
when the electron jumps from state B to C. Wavelength of the radiations emitted when an electron jumps from state A to B will be
Options
A.2000 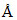
B.3000 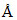
C.4000 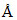
D.6000 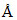
Solution
EA → C = EA → B + EB → C
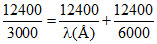 ⇒ λ = 6000
⇒ λ = 6000 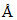
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
A stream of electrons from a heated filament was passed between two charged plates kept at a potential difference V esu....An α-particle having kinetic energy 4.0 MeV is projected towards tin nucleus (Z = 50). Select the correct information(s)...The number of orbitals of g-type...The atomic nucleus contains :...Consider the compound given belowH2C = CH - CH = CH - CH2 - OH The number of sp2 hybridised atoms is...