ThermodynamicsHard
Question
At a constant volume the specific heat of a gas is 0.075 and its molecular weight is 40. The gas is :
Options
A.Monoatomic
B.Diatomic
C.Triatomic
D.None of the aboveNone of the above
Solution
We know that, Molar heat capacity at constant volume,
Cv = Specific heat at constant volume × Mol. wt.
= 0.075 × 40 = 3.0 cal
∵ Cp - Cv = R
or Cp = R + Cv 2 + 3 = 5
Now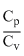 = γ, ∴ γ =
= γ, ∴ γ = 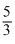 = 1.66
= 1.66
This value shows that the gas is mono atomic.
Cv = Specific heat at constant volume × Mol. wt.
= 0.075 × 40 = 3.0 cal
∵ Cp - Cv = R
or Cp = R + Cv 2 + 3 = 5
Now
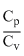 = γ, ∴ γ =
= γ, ∴ γ = 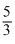 = 1.66
= 1.66This value shows that the gas is mono atomic.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A piston filled with 0.04 mol of an ideal gas expands reversibly from 50.0 mL to 375 mL at a constant temperature of 37....Which of the following processes takes place with decrease of entropy ?...Internal energy does not include:...Equal volumes of two monoatomic gases, A and B, at same temperature and pressure are mixed. The ratio of specific heats ...Which of the following statement(s) is/are false ?...