ThermodynamicsHard
Question
Equal volumes of two monoatomic gases, A and B, at same temperature and pressure are mixed. The ratio of specific heats (Cp/Cv) of the mixture will be
Options
A.1.67
B.0.83
C.1.50
D.3.3
Solution
Cp of the mixture = 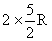
Cv of the mixture =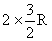
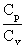 of the mixture = 1.67
of the mixture = 1.67
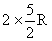
Cv of the mixture =
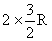
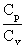 of the mixture = 1.67
of the mixture = 1.67Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
An ideal gas undergoes a process in which its pressure and volume are related as PV n = constant, where n is a constant....Which of the following would be expected to have the largest entropy per mole?...If x and y are arbitrary extensive variables, then...The work involved (w) in an isothermal expansion of n moles of an ideal gas from an initial pressure of ‘P’ atm to final...The work done on one mole of an ideal gas in the reversible process: PV3 = constant, from (1 atm, 300 K) to 2 2 atm is...