Mole ConceptHard
Question
Volume of a gas at NTP is 1.12 × 10-7cc. The number of molecule in it is :
Options
A.3.01 × 1012
B.3.01 × 1018
C.3.01 × 1024
D.3.01 × 1030
More Mole Concept Questions
Vapour pressure of chloroform (CHCl3) and dichloromethane (CH2Cl2) at 25oC are 200 mm Hg and 41.5 mm Hg respectively. Va...CH3-CO-CH3(g) ⇋ CH3-CH3 (g) + CO(g)Initial pressure of CH3COCH3 is 100 mm. When equilibrium is set up mole fractio...The absolute configuration of the two chiral centres in the following molecule are...In which of the following reactions, increase in the volume at constant temperature does not affect the number of moles ...Which of the following statement is not true about H2O molecule :-...
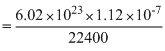
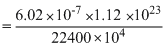 = 3.01 × 1012
= 3.01 × 1012