Mole ConceptHard
Question
CH3-CO-CH3(g) ⇋ CH3-CH3 (g) + CO(g)
Initial pressure of CH3COCH3 is 100 mm. When equilibrium is set up mole fraction of CO(g) is 1/3, hence Kp is :-
Initial pressure of CH3COCH3 is 100 mm. When equilibrium is set up mole fraction of CO(g) is 1/3, hence Kp is :-
Options
A.100 mm
B.50 mm
C.25 mm
D.150 mm
Solution
CH3COCH3(g) ⇋ CH3-CH3(g) + CO (g)
100 0 0
100 - x x x
total = 100 + x
Now mole fraction of CO =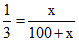
x = 50
CH3COCH3(g) ⇋ CH3-CH3(g) + CO(g)
50 50 50
Kp =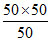 = 50 mm
= 50 mm
100 0 0
100 - x x x
total = 100 + x
Now mole fraction of CO =
x = 50
CH3COCH3(g) ⇋ CH3-CH3(g) + CO(g)
50 50 50
Kp =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
A gaseous mixture contains oxygen and nitrogen in the ratio of 1 : 4 by weight. Therefore, the ratio of their number of ...Mole fraction of the component A in vapour phase is x1 and mole fraction of component A in liquid mixture is x2 (PoA = v...A 2 m long tube closed at one end is lowered vertically into water until the closed end is flushed with the water surfac...Which of the following statement is not true about H2O molecule :-...Molecular mass of dry air is...