Mole ConceptHard
Question
The weight of a single atom of oxygen is :
Options
A.1.057 × 1023 g
B.3.556 × 1023 g
C.2.656 × 1023 g
D.4.538 × 1023 g
Solution
Weight of single a tom of oxygen
=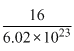 2.656 × 10-23g
2.656 × 10-23g
=
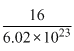 2.656 × 10-23g
2.656 × 10-23gCreate a free account to view solution
View Solution FreeMore Mole Concept Questions
Time required to deposit one milli-mole of aluminium metal by the passage of 9.65 amperes through aqueous solution of al...One mole of the aquous solution of complex CoCl3.4NH3 passess through anion exchange resine and resulting solution treat...A bag contains 0.32 g of oxygen. The same volume of an unknown gas A under similar condition of temperature and pressure...Total charge required for the oxidation of two moles Mn3O4 into MnO4-2 in presence of alkaline medium is :-...If 0.50 mole of BaCl2 is mixed with 0.20 mole of Na3 PO4, the maximum number of moles of Ba3(PO4)2 that can be formed is...