Mole ConceptHard
Question
A bag contains 0.32 g of oxygen. The same volume of an unknown gas A under similar condition of temperature and pressure is present in another container having mass 0.26 gm. If C and H are in 1 : 1 ratio in compound. The molecular formula of the compound is -
Options
A.C2H2
B.C4H4
C.C3H4
D.CH
Solution
Moles of O2 in bag A = 0.32/32 = 0.01 mol
Same volume of unknown gas = = 0.01mol
0.01 mol of unknown gas A = 0.26 g
1 mol of unknown gas A =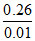 = 26 g
= 26 g
Mwt of unknown gas A = 26 g
Empirical formula = CH
Empirical formula weight = 12 + 1 = 13 g
n =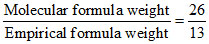 = 2
= 2
Molecular formula = (CH)2 = C2H2.
Same volume of unknown gas = = 0.01mol
0.01 mol of unknown gas A = 0.26 g
1 mol of unknown gas A =
Mwt of unknown gas A = 26 g
Empirical formula = CH
Empirical formula weight = 12 + 1 = 13 g
n =
Molecular formula = (CH)2 = C2H2.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The energy of activation is :-...The pair of species having identical shapes for molecules of both species is...Density of a 2.05 M solution of acetic acid in water is 1.02 g/mL. The molality of the solution is...Select the correct statement about the given complex-...The vapour density of the gas X is twice that of the gas Y. If the molecular weight of the gas X is 4 g/mole, the molecu...