AIPMT | 2014Mole ConceptHard
Question
In the Kjeldahl′s method for estimation of nitrogen present in a soil sample, ammonia evolved from 0.75 g of sample neutralized 10 mL of 1 M H2SO4. The percentage of nitrogen in the soil is
Options
A.37.33
B.45.33
C.35.33
D.43.33
More Mole Concept Questions
The mass of CO2 that must be mixed with 20 g of oxygen such that 27 ml of a sample of the resulting mixture would contai...An amount of 1 mole of calcium cyanamide and 1 mole of water are allowed to react. The number of moles of ammonia produc...The energy of activation is :-...A quantity of 2.0 g of a triatomic gaseous element was found to occupy a volume of 448 ml at 76 cm of Hg and 273 K. The ...Assume that the atomic mass of oxygen is 7. A sample of 11 g of an oxide of uranium contains 10 g of uranium. Which of t...
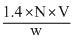
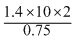 = 37.33%
= 37.33%