AIPMT | 2014Chemical BondingHard
Question
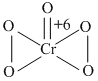
In acidic medium, H2O2 - changes Cr2O7-2 to CrO5 which has two (-O-O-) bonds. Oxidation state of Cr in CrO5 is
Options
A.+5
B.+3
C.+6
D.-10
More Chemical Bonding Questions
The pair of species with the same bond order is...Which of the following is para-magnetic ?...In [Ag(CN2)]-, the number of π bonds is :...The mechanism for the overall reaction isA2 + B → CA2 → 2A (slow)2A + B → C (fast)If a catalyst D chan...Which pair have identical bond order :-...