Chemical BondingHard
Question
The pair of species with the same bond order is
Options
A.O22-, B2
B.O2+, NO+
C.NO, Co
D.N2O2
Solution
O22- (8 + 8 + 2 = 18) = σls2, σ*ls2, σ2s2, σ*2s2, σ2p2z , π2p2x ,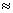 π2p2y , π* 2p2x
π2p2y , π* 2p2x 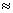 π* 2p2y
π* 2p2y
Bond order (BO)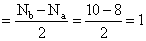
B2 (5 + 5 = 10) = σls2, σ*ls2, σ2s2, σ*2s2, π2p1x ,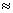 π2p2y
π2p2y
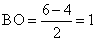
Thus,O22- and B2 have the same bond order.
Note BO of O22- = 2.5, NO- = 2, NO = 2.5, CO = 3, N2 = 3 and O2 = 2
 π2p2y , π* 2p2x
π2p2y , π* 2p2x  π* 2p2y
π* 2p2yBond order (BO)
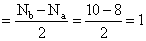
B2 (5 + 5 = 10) = σls2, σ*ls2, σ2s2, σ*2s2, π2p1x ,
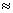 π2p2y
π2p2y 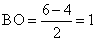
Thus,O22- and B2 have the same bond order.
Note BO of O22- = 2.5, NO- = 2, NO = 2.5, CO = 3, N2 = 3 and O2 = 2
Create a free account to view solution
View Solution Free