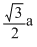AIPMT | 2014Solid StateHard
Question
If a is the length of the side of a cube, the distance between the body centered atom and one corner atom in the cube will be
Options
A.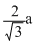
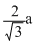
B.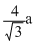
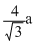
C.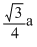
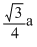
D.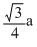
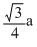
More Solid State Questions
A mineral having the formula AB2, crystallises in the cubic close - packed lattice, with the A atoms occupying the latti...Lithium has a bcc structure. Its density is 530 kg m-3 and its atomic mass is 6.94 g mol-1. Calculated the edge length o...What is the formula of the magnetic oxide of cobalt, used in recording tapes, that crystallizes with cobalt atoms occupy...In a compound, oxide ions are arranged in cubic close packing arrangement. Cations A occupy one-sixth of the tetrahedral...An ionic solid of XY type having anions in CCP lattice and cations in the octahedral voids.Let a be the edge length of a...