Solid StateHard
Question
In a compound, oxide ions are arranged in cubic close packing arrangement. Cations A occupy one-sixth of the tetrahedral voids and cations B occupy one-third of the octahedral voids. The formula of the compound is
Options
A.A2BO3
B.AB2O3
C.A2B2O2
D.ABO3
Solution
no. of oxide ions = 4
no. of A particles =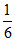 × 8 =
× 8 = 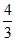
no. of B particles =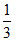 × 4 =
× 4 = 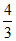
so formula is A4/3 B4/3O4 or ABO3
no. of A particles =
no. of B particles =
so formula is A4/3 B4/3O4 or ABO3
Create a free account to view solution
View Solution FreeMore Solid State Questions
Which of the following solid substance(s) will have the same refractive index when measured in different directions?...In which of the following crystals, alternate tetrahedral voids are occupied ?...The correct statement(s) for the packing of identical spheres in two dimensions is/ are......An ionic crystalline solid MX3 has a cubic unit cell. Which of the following arrangement of the ions is consistent with ...