Atomic StructureHard
Question
When an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas, is :-
Options
A.2/5
B.3/5
C.3/7
D.5/7
Solution
Fraction of supplied energy which increases the internal energy is given by
f =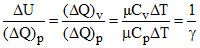
For diatomic gas γ =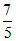 ⇒ f =
⇒ f = 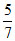
f =
For diatomic gas γ =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Consider α- particle, β - particle and γ- particle each having an energy of 0.5 MeV. In increasing order ...Which one of the following is used as a moderator in reaction ?...When an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which in...A light emitting diode (LED) has a voltage drop of 2 volt across it and passes a current of 10 mA.When it operates with ...The transition from the state n = 3 to n = 1 in a hydrogen like atom results in ultraviolet radiation.Infrared radiation...