ThermodynamicsHard
Question
When a system is taken from a state i to f along the path iaf (as shown in the figure). Q = 50 cal and W = 20 cal; along path ibf, Q = 36cal.
(i) What is W along path ibf ?
(ii) If W = 13 cal for path fi, what is Q for path fi ?
(iii) Take Eint,i = 10 cal than what is Eint,f ?
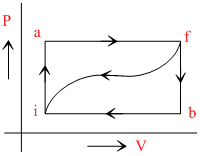
(i) What is W along path ibf ?
(ii) If W = 13 cal for path fi, what is Q for path fi ?
(iii) Take Eint,i = 10 cal than what is Eint,f ?

Options
A.30, 20, 40 cal
B.6, -43, 40cal
C.10, -20, 30cal
D.15, 35, 25cal
Solution
For path iaf
Q = 50
W = 20
According ot I law thermodynamics,
dQ = dU + dW
or dU = dQ - dW = 50 - 20 = 30 cal.
(i) For path iaf
Q = 36 cal.
W = ?
dU = 30 cal (since internal energy depemds only on the initial and final position of the system).
∴ W = Q - dU = 36 - 30 = 6 cal.
(ii) W = - 13cal.
dU = - 30 cal.
Q = ?
∴ Q = dU + W = - 43 cal.
Q = 50
W = 20
According ot I law thermodynamics,
dQ = dU + dW
or dU = dQ - dW = 50 - 20 = 30 cal.
(i) For path iaf
Q = 36 cal.
W = ?
dU = 30 cal (since internal energy depemds only on the initial and final position of the system).
∴ W = Q - dU = 36 - 30 = 6 cal.
(ii) W = - 13cal.
dU = - 30 cal.
Q = ?
∴ Q = dU + W = - 43 cal.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Cp and Cv are the specific heats of 1 gm mole of a gas constant pressure and constant volume respectively. The ratio of ...Two cylinders A and B fitted with piston contain equal amounts of an ideal diatomic gas at 300 K. The piston of A is fre...Two bodies A and B have thermal emissivities of 0.01 and 0.81 respectively. The outer surface area of the two bodies are...A carnot′s engine works between temperature 772oC and 27oC, the efficienty of the engine is :...Consider two process A and B on a system as shown in the figure let ᐃW1 and ᐃW2 be the work done by the syst...