ThermodynamicsHard
Question
Two cylinders A and B fitted with piston contain equal amounts of an ideal diatomic gas at 300 K. The piston of A is free to move, while that B is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in A is 30 K, then the rise in the temperature of the gas in B is
Options
A.30 K
B.18 K
C.50 K
D.42 K
Solution
A is free to move, therefore, heat will be supplied at constant pressure
∴ dQA = nCp dTA .......(i)
B is held fixed, therefore, heat will be supplied at constant volume
∴ dQB = nCv dTB .......(ii)
But dQA dQB [Given]
∴ nCp dTA = nCv dTB ∴ dTB =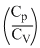 dTA
dTA
= γ(dTA) [γ = 1.4(diatomic)]
(dTA = 30 K)
= (1.4) (30 K)
∴ dTB = 42K.
∴ dQA = nCp dTA .......(i)
B is held fixed, therefore, heat will be supplied at constant volume
∴ dQB = nCv dTB .......(ii)
But dQA dQB [Given]
∴ nCp dTA = nCv dTB ∴ dTB =
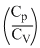 dTA
dTA = γ(dTA) [γ = 1.4(diatomic)]
(dTA = 30 K)
= (1.4) (30 K)
∴ dTB = 42K.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A gas for which γ = 5/3 is heated at constant pressure. The percentage of total heat given that will be used for ex...The process in which the heat given to a system is completely transformed into work is for ddeal gas : -...The average kinetic energy of a gas molecule at 27oC is 6.21 × 10-21J, then its average kinetc energy at 227oC is :...During isothermal, isobaric and adiabatic processes, work done for same change in volume will be maximum for :-...The latent heat of vaporization of water is 2240J. If the work done in the process of vaporization of 1g is 168 J then i...