ThermodynamicsHard
Question
Consider two process A and B on a system as shown in the figure let ᐃW1 and ᐃW2 be the work done by the system in the process A and B respectively then :-
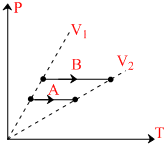

Options
A.ᐃW1 > ᐃW2
B.ᐃW1 < ᐃW2
C.ᐃW1 = ᐃW2
D.Nothing can be said about the relation between ᐃW1 and ᐃW2
More Thermodynamics Questions
The following is wrong in adiabatic process...On mixing equal volumes of a monoatomic gas and a diatomic gas at the same initial pressure and temprature the ratio of ...Two moles of a monoatomic ideal gas is confined in a container and is heated such that its temperature increases by 10oC...An ideal gas heat engine operates in a cornot cycle between 227oC and 127oC. It absorbs 6 kcal at the higher temperature...If the temperature of a black body increases from 7oC to 287oC, then the rate of emission of radiation energy is :...