ThermodynamicsHard
Question
In an adiabatic change, the pressure and temperature of a monoatomic gas are related as P ∝ TC, Where C equals
Options
A.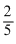
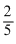
B.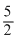
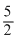
C.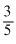
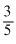
D.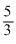
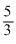
Solution
In adiabatic process, Pγ-1 ∝ Tγ where γ = 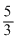
For monoatomic gas ∴ P ∝ Tγ/(γ-1)
∴ C =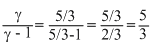
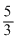
For monoatomic gas ∴ P ∝ Tγ/(γ-1)
∴ C =
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Two cylinders A and B fitted with piston contain equal amounts of an ideal diatomic gas at 300 K. The piston of A is fre...The temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is...At what temperature the molecules of nitrogen will have the same rms velocity as the molecules of oxygen at 127oC...Suppose the sun expands so that its radius becomes 100 times its present radius and its surface temperature becomes half...Three objects colored black, gray and white can withstand hostile conditions upto2800oC. These objects are thrown into a...