ThermodynamicsHard
Question
At what temperature the molecules of nitrogen will have the same rms velocity as the molecules of oxygen at 127oC
Options
A.457oC
B.273oC
C.350oC
D.77oC
Solution
Rms velocity of gas is
Vrms = 1.73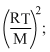
M = molecular mass
For oxygen,M = 16 × 2,
T = 127oC = 127 + 273 = 400 K
For nitrogen, M = 17 × 2, T = ?
⇒ 1.73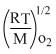 = 1.73
= 1.73 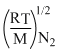
⇒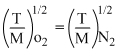
⇒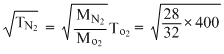
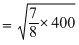
⇒ TN2 =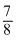 × 400 = 350K
× 400 = 350K
TN2 = 350K - 273K = 77o C
Vrms = 1.73
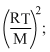
M = molecular mass
For oxygen,M = 16 × 2,
T = 127oC = 127 + 273 = 400 K
For nitrogen, M = 17 × 2, T = ?
⇒ 1.73
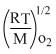 = 1.73
= 1.73 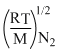
⇒
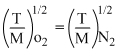
⇒
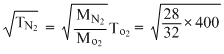
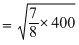
⇒ TN2 =
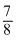 × 400 = 350K
× 400 = 350KTN2 = 350K - 273K = 77o C
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The volume of a metal sphere increases by 0.15 % when its temperature is raised by 24oC.The coefficient of linear expans...In the above question, the curve for which the heat is absorbed from the surroundings is :-...Two monoatomic ideal gases 1 and 2 of molecular masses m1 and m2 respectively are enclosed in separate containers kept a...Temperature of an ideal gas is 300 K. The change in temperature of the gas when its volume changes from V to 2V in the p...A system does 50 J of work under adiabatic condition. In this process :-...