ThermodynamicsHard
Question
Vrma′ Vav and Vmp are root mean squre, average and most probale speeds of molecules of a gas obeying Maxwellian velosity distribution. Which of the following statements is correct ?
Options
A.Vrma < Vav < Vmp
B.Vrma > Vav > Vmp
C.Vmp < Vrma < Vav
D.Vmp > Vrma > Vav
Solution
vrms = 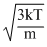 ; vmp =
; vmp = 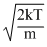
vav =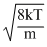
So, vrms > vav > vmp
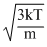 ; vmp =
; vmp = 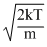
vav =
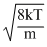
So, vrms > vav > vmp
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
In the diagrams (i) to (iv) of variation of volume with changing pressure is shown. A gas is taken along the path ABCDA....Three objects colored black, gray and white can withstand hostile conditions upto2800oC. These objects are thrown into a...On mixing equal volumes of a monoatomic gas and a diatomic gas at the same initial pressure and temprature the ratio of ...A diatomic ideal gas is used in a Car engine as the working substance. If during the adiabatic expansion part of the cyc...A bullet moving with velocity v collides against wall. consequently half of its kinetic energy is converted into heat. I...