ThermodynamicsHard
Question
In the diagrams (i) to (iv) of variation of volume with changing pressure is shown. A gas is taken along the path ABCDA. The change in internal energy of the gas will be:-
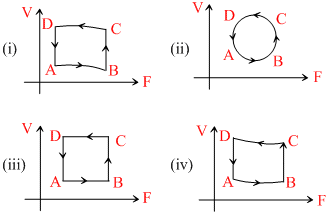

Options
A.Positive in all cases (i) to (iv)
B.Positive in cases (i), (ii) and (iii) but zero in case (iv)
C.Negative in cases (i), (ii) and (iii) but zero in case (iv)
D.Zero in all the four cases
More Thermodynamics Questions
If an ideal flask containing hot coffee is shaken, the temperature of the coffee will :...Which statement is incorrect ?...A black body is at temperature if 2800 K. The energy of radiation emitted by this body with wavelength between 499 nm an...20 litres of hydrogen gas at one atmospheric pressure is compressed at 5 atmospheric pressure, without involving any cha...The intensity of radiation emitted by the sun has its maximum value at a wavelength of 510 nm and emitted by the north s...