ThermodynamicsHard
Question
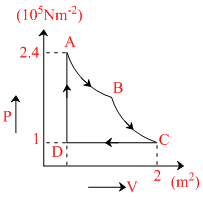
100 mole of an ideal monoatomic gas undergoes the following thermodynamic process as shown in the figure (P - V or Pressure - Volume plots are shown)
A → B : isothermal expansion
B → C : adiabatic expansion
C → D : isobaric compression
D → A : isochoric process
The heat transfer along the process AB is 9 × 104 J. The net work done by the gas during the cycle is (Take R = 8 J. K-1 mole-1)
A → B : isothermal expansion
B → C : adiabatic expansion
C → D : isobaric compression
D → A : isochoric process
The heat transfer along the process AB is 9 × 104 J. The net work done by the gas during the cycle is (Take R = 8 J. K-1 mole-1)

Options
A.- 0.5 × 104 J
B.+ 0.5 × 104 J
C.-5 × 104 J
D.+5 × 104 J
Solution
Wnet = WAB + WBC + WCD + WDA
= 9 × 104 +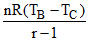 + P(V2 - V1) + 0
+ P(V2 - V1) + 0
TB = TA = 300 K, TC =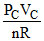
= 9 × 104 +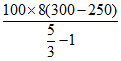 + 105( 1 - 2) + 0
+ 105( 1 - 2) + 0
= 5 × 104 joule
= 9 × 104 +
TB = TA = 300 K, TC =
= 9 × 104 +
= 5 × 104 joule
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Amount of work which can be obtained from 200 cal heat, will be :...An ideal gas is taken around the cycle ABCA as shown in the P - V diagram. The net work done by the gas during the cycle...A black body, at a temperature of 727oC, radiates heat at a rate of 20 cal m-2s-1. When its temperature is raised to 727...Two metallic spheres S1 and S2 are made of the same material and have got identical surface finish. The mass of S1 is th...Most accurate values are :-...