ThermodynamicsHard
Question
Two moles of a monoatomic ideal gas is confined in a container and is heated such that its temperature increases by 10oC. The approximate change in the internal energy is
(R = 8.31J/mole-K)
(R = 8.31J/mole-K)
Options
A.+ 250joules
B.+ 350jouiles
C.-250joules
D.+450joules
More Thermodynamics Questions
The thermal conductivity of a rod 2.What is its thermal resistivity ?...A diatomic ideal gas is heated under constant pressure, the part of heat that increase internal energy of gas is :-...An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume V1 a...One mole of an ideal gas undergoes a process whose molar heat capacity is 4R and in which work done by gas for small cha...″Heat cannot by itself flow from a body at lower temperature to a body at higher temperature″ is a statement of cons...
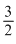 RᐃT
RᐃT